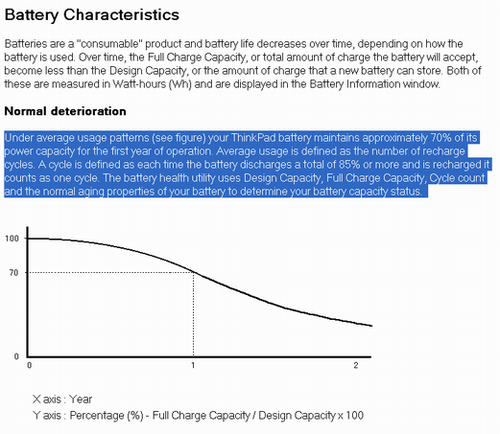
แบตเตอร์รี่แบบ Li-Ion และรวมถึงแบตฯรุ่นใหม่ Li-Polymer ด้วย นั้นจะ นับรอบการชาร์จ (Cycle) ของแบตฯ ของตัวมันเอง ซึ่งรอบการชาร์จของแบต Li-Ion คือ ชาร์จรวมกันแล้ว 85 – 95 % ขึ้นไป (ขึ้นอยู่กับผู้ผลิตด้วย) ถึงจะนับเป็น 1 รอบ ไม่ใช่จำนวนครั้งในการชาร์จ อย่างที่เข้าใจกัน ตัวอย่างเช่น คุณชาร์จไปครั้งแรก ใช้ไปแค่ 20% ซึ่งแบตของคุณในตอนนั้นเหลือ 80% คุณก็ชาร์จไฟเข้าไปใหม่ คุณจะสามารถทำอย่างงี้ไป 5 ครั้ง ถึงจะ นับ 1 รอบ การชาร์จ ผมก็อปมาให้ดูจาก Help ของ Notebook IBM ครับ A cycle is defined as each time the battery discharges a total of 85% or more and is recharged it counts as one cycle. ผมใช้ไปสัก 20 – 30% แล้วชาร์จ ทำแบบนี้ไปราว 3 ครั้ง ตัวเลข Cycle จึงขึ้นมา 1 ครับ (แบตบ้าอะไรก็ไม่รู้ นับจำนวน Cycle ตัวเองได้ด้วย) แต่ถ้าเป็นแบต NiCd หรือ NiMH ก็นับจำนวนครั้งที่ชาร์จเลย ดูได้ดังภาพด้านล่างครับ
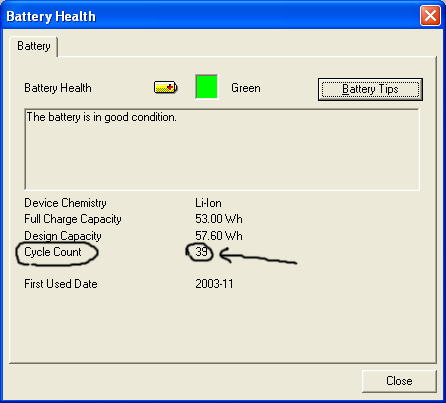
ดูที่ cycle count นะครับ ผมเสียบแบตเข้าออกไม่ต่ำกว่า 200 รอบแล้ว นับจากใช้ notebook ibm ที่ใช้แบต li-ion มา แต่จำนวน cycle count ยังอยู่ที่ 39 อยู่เลยเพราะว่าผมเอาไปเรียนไม่เท่าไหร่กับใช้แบตบ้าง เสียบแบต บาง ไม่เสียบบ้างครับ
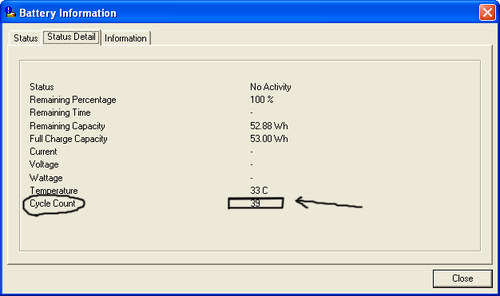
ดูอีกอันนะครับ คงยืนยันได้เป็นอย่างดีครับ ยี่ห้อ ibm คงยืนยันได้ดีนะครับ แถมเป็น li-ion ของ panasonic ที่เป็นฐานการผลิตแบตแบบ oem ใหญ่ให้กับแบตหลายๆ ยี่ห้อรวมทั้ง sony ด้วยครับ ……. ดังภาพด้านล่างสองภาพครับ
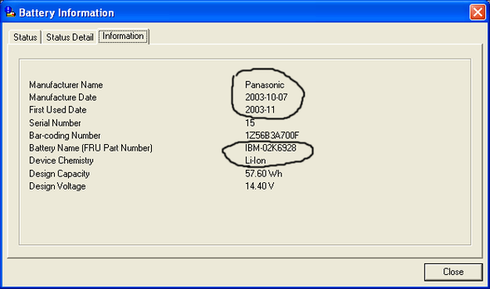
คงต้องบอกว่าตัวแบต Li-ion นั้นระหว่าง Notebook หรือ มือถือ นั้นเหมือนกัน ไม่ต้องบอกก็ดูจะรู้ครับเพราะว่ากรรมวิธีไม่ได้แตกต่างกันเลย มันก็แบบเดียวกันครับ นั้นหมายความว่าการนับย่อมเหมือนกันครับ สารประกอบ และธาตุที่เอามาทำแบตนั้นชนิดเดียวกันโครงสร้างเหมือนกัน ทุกส่วนครับ
หลักการทำงานและสารประกอบทางเคมีครับ
| Lithium is the lightest metallic element and generates a high voltage vs. the standard hydrogen electrode (i.e. -3.045 V). Early attempts used lithium metal in combination with a transition metal oxide or sulphide (e.g. Li/MoS2 the Molicell) intercalation compound. These cells, although exhibiting the high energy densities expected, suffered from two main problems, caused mainly by the lithium anode: limited cycle life and poor safety. The lithium anode caused both the poor safety and limited cycle life. During repeated discharge and charge cycles, the lithium stripping and replatingprocess was not 100% efficient and this created high surface area, particulate lithium which gradually consumes the lithium metal foil anode. The presence of particulate lithium caused increased internal resistance that limits the cycle life. In addition, and more importantly, the particulate lithium creates major safety problems and renders the cell unsafe. The Solution The solution to the so-called ‘lithium metal’ problem was to replace the metal anode with a second intercalation compound which can reversibly intercalate Li+. This material is carbon and this is now used as the active anode material in all commercially available Li-ion cells. Since the anode is carbon, the active material of the cathode must be a compound which already contains Li+ and moreover the Li+ must be easily removed without a change in its molecular (crystal) structure. There are presently two types of compounds which meet this requirement: these are the layered transition metal oxides LiMO2 (where M = Co, Ni or Mn) – examples include LiCoO2 (lithium cobaltite) and LiNiO2 (lithium nickelite) – and the spinel material LiMn2O4. The use of LiCoO2, LiNiO2 and mixed compounds of such as LiNi1-xCoxO2 is protected by several patents owned by AEA Technology. All the major companies which manufacture Li-ion cells have found that LiCoO2 offers superior reversibility, discharge capacity, charge/discharge efficiency and have thus adopted it as the cathode material of choice for small cells used in portable electronics. As Li-ion cells are charged and discharged Li+ ions are transported between their carbon-based anode and their LiCoO2-based cathode, with electrons exchanged as a result of lithium ion insertion (doping) and of lithium ion extraction (undoping). During charging, the cathode is undoped (i.e. the lithium is removed), and the anode, which consist of carbon with a layered structure are doped (i.e. lithium ions are inserted). During discharge (when electrical energy is spontaneously released) lithium is removed from the carbon layers of the anode and inserted into the layers of the cathode compound. When Li-ion cells are first charged, lithium ions are transferred from the layers of the lithium cobaltite to the carbon material which forms the anode. This is illustrated below. 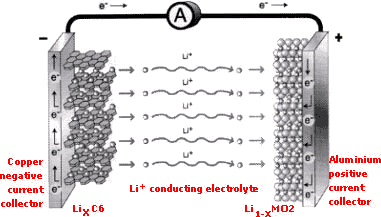 initial charging LiCoO2 + 6C –> Li1-xCoO2 + LixC6 Subsequent discharge and charge reactions are then based on the motion of lithium ions between anode and cathode. discharging Li1-xCoO2 + LixC <—-> Li1-x+dxCoO2 + Lix-dxC charging In order to achieve a high energy density, the capacity of the carbon anode must be as high as possible. To this end, carbon materials with large lithium ion doping capacities are required and the stoichiometric LiC6 lithium-graphite interlated compound composition (corresponding to 372 mA h g-1) were selected. Anode Technology At present, there are three kinds of carbon which are used in the anode of Li-ion cells: Graphite types – highly structured Coke types – less structured but easily transformed into graphite by heating Non-graphitizable (hard) carbon types – highly disordered. Of these carbon types Sony Energytec originally adopted a non-graphitizable (hard) carbon for use in the anode. In contrast, the other leading Li-ion cell manufacturers such as Sanyo, Matushita (Panasonic) and Japan Storage Battery Co., Ltd. (JSB) have adopted a graphitic type carbon for the anode. Although the stored capacity of both graphite and hard carbon cells is similar, the average discharge voltage of the graphite cell (3.7 V) is slightly higher than for hard carbon (3.6 V). The delivered energy of the graphite technology is therefore higher for the same cell capacity due to its flatter discharge characteristic. Recently, non-carbon active anode materials consisting of amorphous tin composite oxide (ATCO) materials have been used by Fujifilm Celltec. These materials have advantages in bulk density and reversible specific capacity compared to graphite. However, the major disadvantage with these materials is a large irreversible capacity that has to date limited their successful introduction in to commercial Li-ion products. อ้างอิง : http://www.agmbat.co.uk/liiontechnology.html |
ซึ่งถ้าปิดหรือเปิดเครื่องหรืออุปกรณ์ระหว่างชาร์จนั้นอันไหนได้ผลดีกว่ากัน อันนี้ผมไม่ขอสรุปเพราะว่ายังไม่มีการทดสอบและทดลองที่แน่ชัดครับ อันนี้คงแล้วแต่สะดวกมากกว่าครับ แล้วอีกอย่างถึงแม้แบตเราจะไม่ได้ทำการชาร์จเลยเป็นเวลานานก็ตามแบตก็จะเสื่อมไปเองภายในเวลา 3 – 5 ปีครับ อันเนื่องมากจากการทำงานของสารเคมีภายในที่หมดคุณภาพไปครับ หรืออาจจะเป็นเพราะข้อจำกัดของสารประกอบและกรรมวิธีของมันเองมากกว่าครับ อันนี้ผมไม่ขอตอบแน่ชัดเพราะว่ายังไม่มีรายงานใดๆ ออกมาครับจึงสรุปได้ไม่เต็มปากครับ แต่ที่สังเกตก็เป็นเช่นนั้นครับ ใช้ไม่ใช้ก็มีอายุเท่ากันแต่ใช้แล้วเนี่ยมันจะสั้นกว่า แต่ก็ไม่ต่างกันมากนักขึ้นอยู่กับปัจจัยหลายๆ อย่างเช่น
- การใช้เครื่องชาร์จที่ได้รับไฟฟ้าที่นิ่งๆ คือการได้รับไฟฟ้าที่ไม่มีไฟตกไฟเกินไฟกระฉาก ครับ อันนี้มีผล ต่อการชาร์จไฟที่มีคุณภาพ 10 – 20% ครับ
- อุณหภูมิในระหว่างการชาร์จ หรือประจุไฟควรประจุที่อุณภูมิปกติ และไม่มีความชื่นมากนักเพราะจะทำให้การถ่ายเทความร้อนทำได้ยากขึ้น
- ขั้วแบตและขั้วส่วนของเสียบสายชาร์จนั้นต้องมีการส่งผ่านไฟที่สม่ำเสมอ เพราะว่าทำให้การประจุไฟหรือการชาร์จเป็นไปอย่างราบรื่นและได้ผลที่ดี
- การหลีกเลี่ยงการทำแบตตกพื้นเพราะจะทำให้หน้าสัมผัสภายในเสียหรือหลุดได้โดยที่เราไม่รู้ รวมถึงทำให้สารประกอบต่างๆ รั้วไหลได้ (เป็นต้นเหตุให้ระเบิดได้)
- ควรใช้แบตอย่างถูกต้องตามแบบสารประกอบนั้นๆ เช่น NiCd ให้ใช้หมดก่อนแล้วชาร์จ NiMH , Li-ion , Li-Poly ลักษณะการใช้งานคล้ายมาก จะชาร์จตอนไหนก็ชาร์จเพียงแต่ NiMH นั้นยังมี memory effect ซึ่ง NiMH นั้นเป็นแบตที่เป็นต้นแบบของ Li-ion เลยก็ว่าได้เพราะว่าเอาแก้ไขส่วนของ memory effect ของ NiCD โดยเฉพาะครับ แต่ว่า Li-ion ทำได้ดีกว่า ส่วน Li-ion กับ Li-Poly นั้นแทบจะไม่มีหรือไม่มีเลย
- การชาร์จในตอนแรกที่ได้รับแบตมานั้น NiCD , NI-HM นั้นใช้ชาร์จ 12 – 14 ชม. 3 ครั้งทุกครั้งใช้แบตให้หมด เพื่อเป้นการกระตุ้นธาตุ Ni ครับ ส่วน Li-ion และ Li-Poly นั้นไม่ต้องครับ แค่ทำให้มันเต็มหรือชัวช์ๆ ก็ 3 ครั้งแรกชาร์จสัก 6 ชม. ก็พอครับ แต่ Li-ion อย่าทำให้แบตหมดเกลี้ยงเป็นอันขาดนะครับ เพราะจะทำให้แบตเสียได้ ส่วน Li-Poly นั้นแก้ไขส่วนนี้มาแล้ว และเป็นแบตที่มีน้ำหนักเบากว่า Li-ion ครับ
- หวังว่าคงเข้าใจพอสมควรแล้วนะครับ ลองหาอ่านได้จากหนังสือ แบตเตอร์รี่ของ Se-ed ครับผมจำได้ว่าการ สร้าง NiMH นั้นสร้างมาเพื่อลบจุดด้อยเรื่อง memory effect ของ NiCD ครับแต่ว่าไม่มากพอซึ่งมีบ้างแต่ไม่มีเท่าครับแต่ได้ความจุที่มากกว่า NiCD มากเลยนั้นคือสิ่งที่ดีของ NiMH ที่ดี แต่ด้อยตรงที่ NiCD นั้นคายประจุได้สม่ำเสมอและเที่ยงตรงมากที่สุดในแบตที่ชาร์จใหม่ได้ครับ …………… ทุกอย่างมีข้อดีและข้อเสียของมันครับ …………
[Update 22/03/2004 = แหล่งความรู้เพิ่มเติม]
- NEW TECHNOLOGY BATTERIES GUIDE, NIJ Guide 200-98 จาก U.S. Department of Justice, Office of Justice Programs, National Institute of Justice
[Update 2/12/2006 = แหล่งความรู้เพิ่มเติม]
ขอบคุณ สำหรับความรู้ ดีดี ค่ะ
ขอบคุณครับ สงสัยมานานแล้ว
ขอบคุณมากครับ
เป็นข้อมูลที่ดีมากเลยครับ…..ขอบคุณๆ
อาตมาใช้เครื่อง notebook ของ acer ซื้อมาใหม่และใช้เครื่องได้ประมาณ ๘ เดือน ประสิทธิภาพในการใช้ของแบตหลังจากถอด Adapter ออก ใช้ได้ประมาณ ๔๐ นาที อาตมาเลยคิดว่าต้องลบประจุให้หมดแล้วลองชาร์จใหม่ ก็เลยใช้งานจนแบตเตอรี่ดับไปเอง พอเสียบสาย Adapter แล้วเปิดเครื่อง สัญญลักษณ์ที่ Task Bar ก็แสดงรูปชาร์จแบตเตอรี่ พอคลิกไปเพื่อจะดูการชาร์จ มีเครื่องหมายขีดสีแดงที่ Charging ก็ชาร์จไฟจนกระทั่งเต็ม ก็ดึงสาย Adapter ออก ลองไปคลิกที่รูปแบตเตอรี่อีกครั้งที่ Task bar ปกติใน Power Meter จะแสดงเวลาที่ยังใช้งานได้อยู่ แต่ตอนนี้มันหายไปแล้ว พอนำ Mouse ไปชี้ที่รูปแบตเตอรี่ที่ task bar ปกติจะบอกเวลาเลย แต่ตอนนี้บอกเพียงว่า 100% remaining (ไม่แสดงเวลาที่ใช้แบตเตอรี่ได้) อาการอย่างนี้แสดงว่าแบตเตอรี่เสียหรือเปล่า แล้วจะมีวิธีไหนที่จะทำให้แบตแสดงรายละเอียด เวลาที่เหลืออยู่ที่สามารถใช้แบตเตอรี่ได้อีกหรือไม่ หรือจะมีวิธีใดที่จะทำให้สมรรถภาพแบตเตอรี่กลับมาใช้งานได้ดีอีกหรือไม่ ชอบตอบหน่อยนะ
เจริญพร
ลืมบอกไปว่าแบตเตอรี่เป็นแบบ Li-ion และถ้าต้องการโหลดโปรแกรมในการควบคุม battery สามารถดาวน์โหลดได้ที่แวบไซต์ไหน
ลอง Notebook BatteryInfo ดูนะครับ
ดาวน์โหลดได้ที่ http://www.softpedia.com/get/Desktop-Enhancements/Other-Desktop-Enhancements/Notebook-BatteryInfo.shtml
ขอบคุณมากครับ กำลังจะชาร์ทแบต li-ion ตอนนี้เหลือประมาณ 40% เลยเข้ามาหาข้อมูลก่อนว่าต้องใช้ให้หมดเลยหรือเปล่า สรุปว่าชาร์ทได้เลย
ขอบคุณมากครับได้ความรู้อีกแล้วเรา ^^ ดีมากๆคัรบ
เครื่อง Com ผม (IBM)เป็นแบตแบบ Li-Ion ถ้าหากว่าผม เสียบ Adapter ไว้ตลอดเวลา ก็แสดงว่า แบตผมไม่ได้ถูกใช้งาน หรือพูดง่ายๆว่าไม่ได้ถูกนับเป็น Cycle ใช่หรือไม่ครับ
พอดีผมเคยใช้แบตฯจนหมดมันจะทำให้แบตฯเสื่อมรึเปล่าและจะมีวิธีแก้ไขอย่างไรครับ แบตฯ li-ion ครับ เพิ่งซื้อมาได้ 3 อาทิตย์เอง (หรือจะส่งเครมประกันเลยครับ)รบกวนช่วยตอบด้วบครับ ตอนนี้แบตฯอยู่ได้ชั่วโมงเดียวครับ
ถ้าการชาร์ทครั้งแรก ชาร์ท Li-Polymer ไปแค่ 1.5 ชั่วโมงก็เต็ม แล้วถอดออก แต่ไม่แน่ใจจึงชาร์ทใหม่ แต่มันขึ้นว่าเเบตเต็มแล้ว
..ก็ยังคงชาร์ทต่อไป เพราะร้านที่ซื้อมาบอกว่า ต้องชาร์ทครั้งแรก 10-12 ชม. …จะไม่มีผลต่อการทำให้เเบตเสื่อมใช่ไม๊คะ
งงอ่ะขร๊ะ!!~
แหะๆ!!~
^^!
จริงหรือค่ะที่ว่าการใช้ไฟบ้าน เราสามารถถอดแบตเตอรี่ออกได้
ทำให้ยืดอายุการใช้งานของ โน๊ตบุ้คได้
ไม่มีความรู้เรื่องนี้เลยจริง ๆ ถ้าจริงจะได้ลองทำดู